Intellipharmaceutics Announces Fiscal Year 2020 Results
TORONTO, ON / ACCESSWIRE / March 1, 2021 / Intellipharmaceutics International Inc. (OTCQB:IPCIF) and (TSX:IPCI) (“Intellipharmaceutics” or the “Company”), a pharmaceutical company specializing in the research, development and manufacture of novel and generic controlled-release and targeted-release oral solid dosage drugs, today reported the results of operations for the year ended November 30, 2020. All dollar amounts referenced herein are in United States dollars unless otherwise noted.
- On July 2, 2020 the Company had announced that the parties in the cases, numbers 17-cv-392-RGA, 18-cv-404-RGA and 20-cv-515-RGA (the “Litigations”) between Purdue Pharma L.P. et al (“Purdue’) and Intellipharmaceutics entered into a stipulated dismissal of the Litigations. The stipulated dismissal, which was subject to approval by the bankruptcy court presiding over Purdue Pharma’s pending chapter 11 cases, provides for the termination of patent infringement proceedings commenced by Purdue against the Company in the United States District Court for the District of Delaware in respect of the Company’s New Drug Application (“NDA”) filing for Aximris XRTM with the U.S. Food and Drug Administration (“FDA”). The stipulated dismissal also provides that (i) for a thirty (30) day period following a final approval of the Company’s Aximris XRTM NDA the parties will attempt to resolve any potential asserted patent infringement claims relating to the NDA and (ii) if the parties fail to resolve all such claims during such period Purdue Pharma will have fifteen (15) days to pursue an infringement action against the Company. The terms of the stipulated dismissal agreement are confidential.
- On July 28, 2020 the United States District Court for the District of Delaware signed the stipulations of dismissal into order thereby dismissing the claims in the three cases without prejudice. In consideration of the confidential stipulated dismissal agreement and for future saved litigation expenses, Purdue has paid an amount to the Company.
- On April 9, 2020, we announced an update on timing of the release of our first quarter financial results for the three months ended February 29, 2020. The Canadian Securities Administrators had announced temporary relief from certain regulatory filings required to be made on or before June 1, 2020 by reporting issuers in Canada, in view of the recent COVID-19 developments and the impact on market participants. The blanket relief provided a 45-day extension for periodic filings, including financial statements and management’s discussion and analysis. We relied on this 45-day extension period provided under the blanket relief for the filing of our interim financial statements for the three months ended February 29, 2020 and the related MD&A. The Company filed its first quarter results for the three months ended February 29, 2020 on May 29, 2020, within the period of extension.
- On February 5, 2020, we announced the resignation of Greg Powell, our former Chief Financial Officer, for personal and family reasons. Pending the hiring of a replacement for Mr. Powell, the functions of Chief Financial Officer are being carried out by our President and former Chief Financial Officer, Dr. Amina Odidi. Fazayill Shaideen, who has been our Controller for the past 8 years, will continue to handle accounting activities.
- On January 15, 2020, at a joint meeting of the Anesthetic and Analgesic Drug Products Advisory Committee and Drug Safety and Risk Management Advisory Committee (“Advisory Committees”) of the FDA to discuss our NDA for Aximris XR™, abuse-deterrent oxycodone hydrochloride extended-release tablets, the Advisory Committees voted 24 to 2 against the approval of our NDA for Aximris XR™ for the management of pain severe enough to require daily, around-the-clock, long-term opioid treatment and for which alternative treatment options are inadequate. We expect the FDA to take action on our application, on completion of their review of the NDA.
Results of Operations
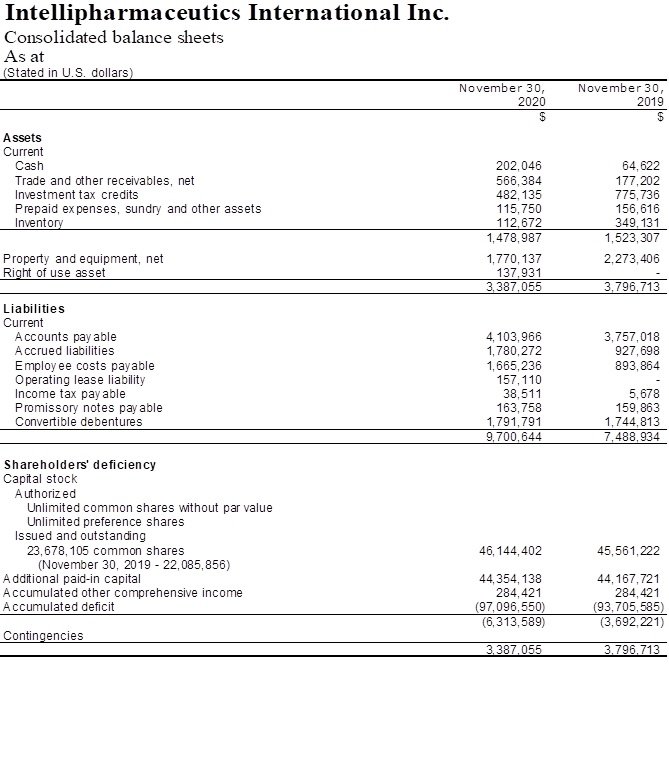
The Company recorded a net loss for the year ended November 30, 2020 of $3,390,965 or $0.14 per common share, compared with a net loss of $8,084,646 or $0.37 per common share for the year ended November 30, 2019. The lower net loss for the year ended November 30, 2020 is attributed to an increase in licensing revenues from commercial sales of generic Focalin XR®, other income received through a stipulated dismissal agreement, as well as a decrease in administrative expenses related to lower professional and legal fees and research and development (“R&D”) expenses related to decreased third party consulting fees, lower expenses related to biostudies and the reduction in number of R&D employees.
The Company recorded revenues of $1,401,517 for the year ended November 30, 2020 versus $3,480,516 for the year ended November 30, 2019. The revenues for the year ended November 30, 2020 consisted solely of licensing revenues from commercial sales of generic Focalin XR® under the Par agreement. The decrease in revenues in the year ended November 30, 2020 compared to the year ended November 30, 2019 is primarily due to the recognition of $2,366,485 from up-front fees in fiscal 2019, resulting from early termination of the Mallinckrodt agreement on August 12, 2019 as compared to the original ten-year term. Beginning in early 2018, we began to see a significant impact on the revenue from sales of generic Focalin XR from aggressive pricing by competitors, resulting in a marked increase in gross-to-net deductions such as wholesaler rebates, chargebacks and pricing adjustments which continues to date. While the gross-to-net deductions fluctuate on a quarter over quarter basis, profit share payments for the 2020 fiscal year have improved over the same period in 2019.
Expenditures for R&D for the year ended November 30, 2020 were lower by $3,091,776 compared to the year ended November 30, 2019. The decrease is due to a reduction in R&D staff, decrease in material purchases and patent and litigation expenses, lower third party consulting fees and a decrease in expenses related to biostudies.
Selling, general and administrative expenses were $2,147,432 for the year ended November 30, 2020 in comparison to $4,167,801 for the year ended November 30, 2019, resulting in a decrease of $2,020,369. The decrease is mainly due to reduced administrative costs and lower wages and marketing costs.
The Company had cash of $202,046 as at November 30, 2020 compared to $64,622 as at November 30, 2019. The increase in cash was mainly due to the receipt of certain payments under the Purdue stipulated dismissal agreement as well as lower expenditures for R&D and selling, general, and administrative expenses.
As of November 30, 2020, our cash balance was $202,046. We currently expect to meet our short-term cash requirements from quarterly profit share payments from Par and by cost savings associated with managing operating expense levels. If we are able to supply products to our marketing and distribution partner, Tris Pharma, and it achieves sales of our generic Seroquel XR®, generic Pristiq and generic Effexor XR at anticipated rates, then we may satisfy our cash needs with cost-saving measures. We will need to obtain additional funding to further product commercialization activities and the development of our product candidates. Potential sources of capital may include payments from licensing agreements, and/or debt financings and/or new strategic partnership agreements which the Company is actively exploring. The Company has funded its business activities principally through the issuance of securities, loans from related parties and funds from development agreements. There is no certainty that such funding will be available going forward. If conditions permit, we intend to utilize the equity markets and/or debt financing to bridge any funding shortfall. Our future operations are highly dependent upon our ability to source additional capital to support advancing our product pipeline through continued R&D activities and to fund any significant expansion of our operations. Our ultimate success will depend on whether our product candidates receive approval by the FDA or Health Canada or the regulatory authorities of other countries in which our products are proposed to be sold and on and whether we are able to successfully market our approved products. We cannot be certain that we will receive FDA or Health Canada or such other regulatory approval for any of our current or future product candidates, that we will reach the level of sales and revenues necessary to achieve and sustain profitability, or that we can secure other capital sources on terms or in amounts sufficient to meet our needs or at all.
There can be no assurance that we will not be required to conduct further studies for our Aximris XR product candidate, that the FDA will approve any of our requested abuse-deterrence label claims, that the FDA will meet its deadline for review or that the FDA will ultimately approve the NDA for the sale of product candidate in the U.S. market or that the product will ever be successfully commercialized and produce significant revenue for us.
About Intellipharmaceutics
Intellipharmaceutics International Inc. is a pharmaceutical company specializing in the research, development and manufacture of novel and generic controlled-release and targeted-release oral solid dosage drugs. The Company’s patented Hypermatrix™ technology is a multidimensional controlled-release drug delivery platform that can be applied to a wide range of existing and new pharmaceuticals. Intellipharmaceutics has developed several drug delivery systems based on this technology platform, with a pipeline of products (some of which have received FDA approval) in various stages of development. The Company has ANDA and NDA 505(b)(2) drug product candidates in its development pipeline. These include the Company’s Oxycodone ER based on its proprietary nPODDDS™ novel Point Of Divergence Drug Delivery System (for which an NDA has been filed with the FDA), and Regabatin™ XR (pregabalin extended-release capsules).
Cautionary Statement Regarding Forward-Looking Information
Certain statements in this document constitute “forward-looking statements” within the meaning of the United States Private Securities Litigation Reform Act of 1995 and/or “forward-looking information” under the Securities Act (Ontario). These statements include, without limitation, statements expressed or implied regarding our expectations , plans, goals and milestones, status of developments or expenditures relating to our business, plans to fund our current activities, and statements concerning our partnering activities, health regulatory submissions, strategy, future operations, future financial position, future sales, revenues and profitability, projected costs and market penetration and risks or uncertainties arising from the delisting of our shares from Nasdaq and our ability to comply with OTCQB and TSX requirements. In some cases, you can identify forward-looking statements by terminology such as “appear”, “unlikely”, “target”, “may”, “will”, “should”, “expects”, “plans”, “plans to”, “anticipates”, “believes”, “estimates”, “predicts”, “confident”, “prospects”, “potential”, “continue”, “intends”, “look forward”, “could”, “would”, “projected”, “goals” ,”set to”, “seeking” or the negative of such terms or other comparable terminology. We made a number of assumptions in the preparation of our forward-looking statements. You should not place undue reliance on our forward-looking statements, which are subject to a multitude of known and unknown risks and uncertainties that could cause actual results, future circumstances or events to differ materially from those stated in or implied by the forward-looking statements. Risks, uncertainties and other factors that could affect our actual results include, but are not limited to, ,the effects of general economic conditions, securing and maintaining corporate alliances, our estimates regarding our capital requirements, and the effect of capital market conditions and other factors, including the current status of our product development programs, capital availability, the estimated proceeds (and the expected use of any proceeds) we may receive from any offering of our securities, the potential dilutive effects of any future financing, potential liability from and costs of defending pending or future litigation, risks associated with the novel coronavirus (COVID-19) including its impact on our business and operations, our programs regarding research, development and commercialization of our product candidates, the timing of such programs, the timing, costs and uncertainties regarding obtaining regulatory approvals to market our product candidates and the difficulty in predicting the timing and results of any product launches, the timing and amount of profit-share payments from our commercial partners, and the timing and amount of any available investment tax credits, the actual or perceived benefits to users of our drug delivery technologies, products and product candidates as compared to others, our ability to establish and maintain valid and enforceable intellectual property rights in our drug delivery technologies, products and product candidates, the scope of protection provided by intellectual property rights for our drug delivery technologies, products and product candidates, recent and future legal developments in the United States and elsewhere that could make it more difficult and costly for us to obtain regulatory approvals for our product candidates and negatively affect the prices we may charge, increased public awareness and government scrutiny of the problems associated with the potential for abuse of opioid based medications, pursuing growth through international operations could strain our resources, our limited manufacturing, sales, marketing and distribution capability and our reliance on third parties for such, the actual size of the potential markets for any of our products and product candidates compared to our market estimates, our selection and licensing of products and product candidates, our ability to attract distributors and/or commercial partners with the ability to fund patent litigation and with acceptable product development, regulatory and commercialization expertise and the benefits to be derived from such collaborative efforts, sources of revenues and anticipated revenues, including contributions from distributors and commercial partners, product sales, license agreements and other collaborative efforts for the development and commercialization of product candidates, our ability to create an effective direct sales and marketing infrastructure for products we elect to market and sell directly, the rate and degree of market acceptance of our products, delays in product approvals that may be caused by changing regulatory requirements, the difficulty in predicting the timing of regulatory approval and launch of competitive products, the difficulty in predicting the impact of competitive products on sales volume, pricing, rebates and other allowances, the number of competitive product entries, and the nature and extent of any aggressive pricing and rebate activities that may follow, the inability to forecast wholesaler demand and/or wholesaler buying patterns, seasonal fluctuations in the number of prescriptions written for our generic Focalin XR® capsules which may produce substantial fluctuations in revenue, the timing and amount of insurance reimbursement regarding our products, changes in laws and regulations affecting the conditions required by the FDA for approval, testing and labeling of drugs including abuse or overdose deterrent properties, and changes affecting how opioids are regulated and prescribed by physicians, changes in laws and regulations, including Medicare and Medicaid, affecting among other things, pricing and reimbursement of pharmaceutical products, the effect of recent changes in U.S. federal income tax laws, including but not limited to, limitations on the deductibility of business interest, limitations on the use of net operating losses and application of the base erosion minimum tax, on our U.S. corporate income tax burden, the success and pricing of other competing therapies that may become available, our ability to retain and hire qualified employees, the availability and pricing of third-party sourced products and materials, challenges related to the development, commercialization, technology transfer, scale-up, and/or process validation of manufacturing processes for our products or product candidates, the manufacturing capacity of third-party manufacturers that we may use for our products, potential product liability risks, the recoverability of the cost of any pre-launch inventory, should a planned product launch encounter a denial or delay of approval by regulatory bodies, a delay in commercialization, or other potential issues, the successful compliance with FDA, Health Canada and other governmental regulations applicable to us and our third party manufacturers’ facilities, products and/or businesses, our reliance on commercial partners, and any future commercial partners, to market and commercialize our products and, if approved, our product candidates, difficulties, delays or changes in the FDA approval process or test criteria for ANDAs and NDAs, challenges in securing final FDA approval for our product candidates, including our oxycodone hydrochloride extended release tablets product candidate, in particular, if a patent infringement suit is filed against us with respect to any particular product candidates (such as in the case of Oxycodone ER), which could delay the FDA’s final approval of such product candidates, healthcare reform measures that could hinder or prevent the commercial success of our products and product candidates, the risk that the FDA may not approve requested product labeling for our product candidate(s) having abuse-deterrent properties and targeting common forms of abuse (oral, intra-nasal and intravenous), risks associated with cyber-security and the potential for vulnerability of our digital information or the digital information of a current and/or future drug development or commercialization partner of ours, and risks arising from the ability and willingness of our third-party commercialization partners to provide documentation that may be required to support information on revenues earned by us from those commercialization partners. Additional risks and uncertainties relating to us and our business can be found in the “Risk Factors” section of our latest annual information form, our latest Form 20-F, and our latest Form F-1 and F-3 registration statements (including any documents forming a part thereof or incorporated by reference therein), as amended, as well as in our reports, public disclosure documents and other filings with the securities commissions and other regulatory bodies in Canada and the U.S., which are available on www.sedar.com and www.sec.gov. The forward-looking statements reflect our current views with respect to future events and are based on what we believe are reasonable assumptions as of the date of this document and we disclaim any intention and have no obligation or responsibility, except as required by law, to update or revise any forward-looking statements, whether as a result of new information, future events or otherwise.
Trademarks used herein are the property of their respective holders.
Unless the context otherwise requires, all references (i) to “we,” “us,” “our,” “Intellipharmaceutics,” and the “Company” refer to Intellipharmaceutics International Inc. and its subsidiaries and (ii) in this document to share amounts, per share data, share prices, exercise prices and conversion rates have been adjusted to reflect the effect of the 1-for-10 reverse split which became effective on each of Nasdaq and TSX at the open of market on September 14, 2018. The common shares of the Company are currently traded on the OTCQB and the TSX.
Nothing contained in this document should be construed to imply that the results discussed herein will necessarily continue into the future or that any conclusion reached herein will necessarily be indicative of our actual operating results.
The consolidated financial statements, accompanying notes to the consolidated financial statements, and Management Discussion and Analysis for the year ended November 30, 2020 will be accessible on Intellipharmaceutics’ website at www.intellipharmaceutics.com and will be available on SEDAR and EDGAR.
Summary financial tables are provided below.



CONTACT INFORMATION
Company Contact:
Intellipharmaceutics International Inc.
Isa Odidi
Chief Executive Officer
416.798.3001 ext. 102
investors@intellipharmaceutics.com
SOURCE: Intellipharmaceutics International Inc.
View source version on accesswire.com:
https://www.accesswire.com/632469/Intellipharmaceutics-Announces-Fiscal-Year-2020-Results
