PYC Therapeutics Announces Preclinical Data Demonstrating Superior PPMO Delivery to the Brain
NEW YORK and PERTH, Australia, April 12, 2021 /PRNewswire/ — PYC Therapeutics (ASX: PYC), a biotechnology company developing a new generation of precision RNA therapeutics to change the lives of patients with inherited diseases, today announced the results of preclinical research demonstrating the ability of its breakthrough PPMO (Peptide conjugated Phosphorodiamidate Morpholino Oligomer) technology to deliver superior levels of RNA therapeutic to cells throughout the brain.
“PYC’s PPMO technology platform has broad potential to address a range of diseases. While we remain focused on progressing our ocular programs into clinical development, we are pleased to share these early but encouraging data in the CNS,” said Sahm Nasseri, U.S. Chief Executive Officer of PYC Therapeutics. “PYC’s PPMO platform is the result of years of careful design and testing. We have very deliberately applied the technology first to diseases of the eye and are excited to strategically expand to other disease areas.”
Over 50 million people globally suffer from a neurodegenerative disease, with over 3 million people suffering from rarer neurodegenerative disease such as amyotrophic lateral sclerosis (ALS) or Huntington’s disease1. The pharmaceutical market size to serve this patient population is estimated to be worth in excess of $30 billion (USD) per annum2 and this is only anticipated to grow with an aging global population. For the vast majority of these patients, there are no disease modifying therapies available today.
A major barrier to development of precision medicines for neurodegenerative disease has been the poor delivery of these medicines to the brain. Insufficient depth of penetration and therefore inadequate delivery to target cells, has prevented drugs from having meaningful impact on these diseases, without causing significant toxicity. This has been especially true for naked PMOs (i.e., PMOs without a delivery molecule), which have particularly poor uptake in adult mouse models, hampering their otherwise ideal safety, durability and target engagement profile — benefits that set PMOs apart from other RNA therapeutics such as Antisense Oligonucleotides (ASOs)3.
As PYC has been previously able to show in ocular diseases, today’s highly encouraging results demonstrate PYC’s PPMO technology has significant potential to solve this delivery challenge in neurodegenerative diseases. Superior delivery and an excellent short- and long-term safety profile in preclinical models could translate to a higher probability of clinical success and ultimately the creation of truly differentiated and meaningful medicines for patients with significant unmet need. Here, PYC has demonstrated PMO delivery and antisense effect at dosages significantly lower than those reported for delivery of both ASO and other RNA therapeutics4, and using research-grade material that may under-represent the true efficacy of PYC’s PPMOs due to the lower purity (an issue that will be overcome as the Company progresses to higher purity drug preparations for testing in larger animal models).
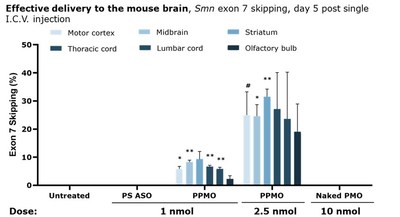
For this study, PYC researchers led by Prof. Sue Fletcher, Chief Scientific Officer of PYC, administered either a PYC PPMO drug, a naked PMO, or a PS ASO drug of the same sequence, via intracerebroventricular injection into mice. On day 5 following treatment, the PPMO demonstrated a dose dependant response across the target areas of the brain including both the motor cortex (focus in ALS) and the midbrain. Further, there was no evidence of efficacy for the ASO or naked PMO at any dose tested in this model. PYC also tested the PMO with additional Cell Penetrating Peptides (CPPs) from the PYC peptide library (each n=1 result) at 1 nmol dosage and observed improved exon-skipping over the current PPMO. While this result is not yet significant due to small sample size, it indicates that PYC’s CPP library contains multiple CPPs that are potentially even more effective for Central Nervous System (CNS) specific delivery.
“Neurodegenerative diseases occur when nerve cells in the brain or spinal cord lose function over time and ultimately die. These conditions affect millions of people worldwide and there are currently no effective treatments,” said Professor Fletcher. “The data reported today are exciting because they demonstrate that we have identified CPPs that show a clear advantage over existing approaches in delivering PMO therapeutics into different regions deep within the brain. We believe this breakthrough result supports the further study of this technology across multiple high unmet need neurodegenerative diseases. Our team is now running experiments to determine the cellular level uptake of the PPMO in the brain – ensuring we deliver to the right cells is the next major de-risking milestone for the CNS PPMO platform.”
“These data support the expansion of our pipeline and discovery efforts to include development of assets aimed at neurodegenerative diseases and conditions of the CNS,” said Mr. Nasseri. “We are actively building out our presence in the U.S. to support translation of our ocular programs into clinical development and we are very excited to continue conducting lead selection work on future CNS candidates. We remain on track to name a candidate targeting a high unmet need neurodegenerative condition in 2021.”
Building off this promising proof-of-concept data, PYC is scaling up discovery efforts to ultimately enable clinical assessment of a promising PPMO drug candidate for a neurodegenerative condition. The Company remains on track to name a CNS drug candidate in 2021 and in parallel, PYC intends to further optimize its delivery technology for CNS administration including assessing both efficacy and safety of PYC’s PPMO technology in larger animals.
About PYC Therapeutics
PYC Therapeutics (ASX: PYC) is a development-stage biotechnology company pioneering a new generation of RNA therapeutics that utilize Cell Penetrating Peptides (CPPs), a revolutionary delivery technology designed to overcome the major challenges of current gene-based therapies. PYC believes its CPP technology provides safer, more effective access for a wide range of potent and precise drug cargoes to the highest value drug targets that exist inside cells. The Company is leveraging its leading-edge science to develop a pipeline of novel therapies with an initial focus on inherited eye diseases for which it has unveiled three preclinical stage assets. PYC’s discovery and laboratory operations are located in Australia and the Company recently launched an expansion into the U.S. for its preclinical, clinical, regulatory and business development operations. For more information, visit pyctx.com, or follow us on LinkedIn and Twitter.
Forward looking statements
Any forward-looking statements in this ASX announcement have been prepared on the basis of a number of assumptions which may prove incorrect and the current intentions, plans, expectations and beliefs about future events are subject to risks, uncertainties and other factors, many of which are outside the Company’s control. Important factors that could cause actual results to differ materially from assumptions or expectations expressed or implied in this ASX announcement include known and unknown risks. Because actual results could differ materially to assumptions made and the Company’s current intentions, plans, expectations and beliefs about the future, you are urged to view all forward-looking statements contained in this ASX announcement with caution. The Company undertakes no obligation to publicly update any forward-looking statement whether as a result of new information, future events or otherwise.
1 GBD 2016 Neurology Collaborators. Global, regional, and national burden of neurological disorders, 1990-2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2019 Parkinson included in ‘rarer disease’ to distinguish from Alzheimer’s.
2 Spinraza peak sales in SMA (Biogen 10-K) scaled by patient populations in other indications. Assumed consistent uptake and pricing (note Sprinraza sold to ~11,000 patients of an estimated 60,000 patients – see 2020 results presentation).
3 See Rigo F, et al. Pharmacology of a central nervous system delivered 2′-O-methoxyethyl-modified survival of motor neuron splicing oligonucleotide in mice and nonhuman primates. J Pharmacol Exp Ther. 2014; 350(1): 46-55.; and Flynn LL, Li R, Aung-Htut MT, et al. Interaction of modified oligonucleotides with nuclear proteins, formation of novel nuclear structures and sequence-independent effects on RNA processing. bioRxiv; 2018.
4 For ASOs in adult mice by I.C.V. see Rigo 2014 cited above at 3; for siRNA see Dicerna Pharmaceuticals Investor presentation, ‘Cell Type-Targeted Therapeutic Oligonucleotides Delivery in the CNS’ available at their company website (accessed April 4, 2021)
5 Nanomoles (nmol) is a measure of how many molecules are dosed, by comparison micrograms (µg) is the weight of molecules dosed. The larger the molecule the higher the µg dose required for the same nmol dose
|
INVESTORS |
MEDIA |
|
Deborah Elson/Matthew DeYoung |
Leo Vartorella |
|
Argot Partners |
Argot Partners |
|
deborah@argotpartners.com |
View original content to download multimedia:http://www.prnewswire.com/news-releases/pyc-therapeutics-announces-preclinical-data-demonstrating-superior-ppmo-delivery-to-the-brain-301266312.html
SOURCE PYC Therapeutics


![Fig 3. Neurodegenerative diseases affecting different regions of the human brain, correlated to the exon skipping induced by PYC’s PPMO in the mouse brain Percent exon skipping achieved after a single 2.5 nmol injection (28 µg dose[5]) depicted in the relevant mouse brain region - Images created using Biorender.com Fig 3. Neurodegenerative diseases affecting different regions of the human brain, correlated to the exon skipping induced by PYC’s PPMO in the mouse brain Percent exon skipping achieved after a single 2.5 nmol injection (28 µg dose[5]) depicted in the relevant mouse brain region - Images created using Biorender.com](https://m.healthtechnologynet.com/wp-content/uploads/2021/04/CNS_Figure_3.jpg)

