Halberd’s COVID-19 Antibody is the Only One In Competitive Tests to Bind to Omicron Variant
Outperforms Approved COVID Antibodies
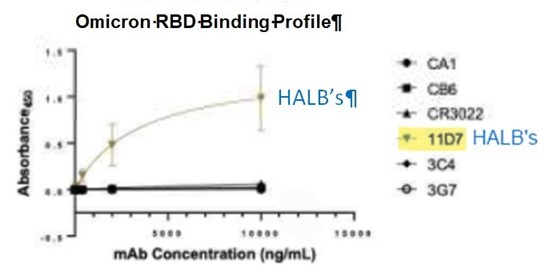
Jackson Center, Pennsylvania–(Newsfile Corp. – March 1, 2022) – Halberd Corporation’s (OTC Pink: HALB) previously developed SARS-CoV-2 antibody demonstrated superior binding capability to the SARS-CoV-2 Omicron variant disease antigen. The Halberd antibody was tested against monoclonal antibodies which are biosimilar to five FDA approved Emergency Use Authorization (EUA) COVID antibodies. The results are shown on the graph below.
On the vertical axis is the Absorbance (a measure of binding capability) of the various monoclonal antibodies (mAbs) to the Omicron variant disease antigen, and the antibody concentration (dose) is shown on the horizontal axis.
Halberd’s antibody is depicted as 11D7, as highlighted on the graph. It reveals that with increased concentrations of the antibody, the binding capability increases. The other five (approved mAbs) have near zero binding, regardless of concentration.
Omicron RBD Binding Profile
To view an enhanced version of this graphic, please visit:
https://orders.newsfilecorp.com/files/7041/115188_omicron%20binding%20profile.jpg
Dr. Mitchell S. Felder, Halberd’s Chief Technology Officer and a board-certified attending neurologist stated, “For an antibody, to be effective in conventional medicine, it must display an affinity for and a binding capability to the target antigen AND have a neutralizing capability against the antigen.”
Dr. Felder continued, “The five competitive monoclonal antibodies that are FDA approved for Emergency Use against Covid, show no binding to the Omicron variant, and thus have no effect against Omicron. If an antibody cannot “find and bind” (binding affinity) to the target antigen, no matter how effective its neutralizing capability is, it is not going to eliminate the target.”
William A. Hartman, Halberd’s Chairman, President & CEO added, “Halberd’s antibody is the only one tested that has demonstrated a strong binding affinity to the Omicron variant of SARS-CoV-2. Although Halberd’s antibody’s neutralizing capability has not yet been tested, Halberd’s patented process doesn’t require any neutralizing capability. We conjugate our antibodies to a metallic nanoparticle, through our patent-pending process, and eliminate the target disease antigen through extracorporeal laser exposure, just as we have done successfully with the 10 neurodegenerative disease antigens.
“This latest COVID development clearly demonstrates Halberd’s superior technical capabilities, whether applied to developing cures for neurodegenerative diseases, as indicated in last week’s announcement, or for developing potential cures for pandemic viruses.”
Hartman continued, “Due to our limited resources, our scientists are focused on development of effective cures for neurodegenerative diseases. As such, we must consider offers for partnerships, sale or licensing of our COVID technology to commercialize its availability to aid patients suffering from Omicron or potentially other variants.”
To view informative videos on Halberd’s work, see:
Dr. Felder interviewed by Alec Torelli (https://www.youtube.com/watch?v=pTM_wQRArOE)
William Hartman interviewed by YSU (https://halberdcorporation.com/wp-content/uploads/2022/02/Alumni-Spotlight-William-A.-Hartman-64.mp4)
To get the latest news on Halberd’s exciting developments, including our ongoing disease eradication accomplishments, subscribe by submitting this form.
(https://halberdcorporation.com/contact-us/)
For more information please contact:
William A. Hartman
w.hartman@halberdcorporation.com
support@halberdcorporation.com
www.halberdcorporation.com
Twitter:@HalberdC
About Halberd Corporation.
Halberd Corporation (OTC Pink: HALB), is a publicly traded company on the OTC Market, and is in full compliance with OTC Market reporting requirements. Since its restructuring in April of 2020, Halberd has obtained exclusive worldwide rights to three issued patents and has filed twenty related provisional, PCT, or utility patent applications to enhance its value to its stockholders and to attract the interests of potential development partners.
Safe Harbor Notice
Certain statements contained herein are “forward-looking statements” (as defined in the Private Securities Litigation Reform Act of 1995). The Company cautions that statements, and assumptions made in this news release constitute forward-looking statements and makes no guarantee of future performance. Forward-looking statements are based on estimates and opinions of management at the time statements are made. These statements may address issues that involve significant risks, uncertainties, estimates made by management. Actual results could differ materially from current projections or implied results. The Company undertakes no obligation to revise these statements following the date of this news release.

To view the source version of this press release, please visit https://www.newsfilecorp.com/release/115188
