Tharimmune Reports Positive Phase 1 Data on TH104, its Lead Therapeutic Candidate, for Chronic Pruritis in Primary Biliary Cholangitis (PBC)

- Phase 1 ex-US trial achieved primary objective of predictable pharmacokinetic profiling with favorable safety and tolerability
- Company expects to engage FDA on Phase 2 trial design and expects topline data in 2024 for chronic pruritis
- First indication for chronic pruritis in PBC where 70% of patients suffer from intractable and debilitating itching with limited treatment options
BRIDGEWATER, NJ / ACCESSWIRE / November 27, 2023 / Tharimmune, Inc. (NASDAQ:THAR) (“Tharimmune” or the “Company”), a clinical-stage biotechnology company developing a portfolio of therapeutic candidates for rare, inflammatory, and oncologic conditions announces positive phase 1 data with TH104, a proprietary transmucosal buccal film which easily adheres to the inside of the mouth. Tharimmune also announced plans on a development timeline for a Phase 2a study in chronic pruritis in primary biliary cholangitis (PBC) patients which will complement the data package the Company expects to discuss with the US FDA and European Regulatory Authorities.
The first Phase 1 trial was a single-dose, single-center, open-label, randomized, 2-way crossover study of TH104 transmucosal buccal film compared to a tablet formulation marketed in Europe and not the United States, with a 14-day washout period involving 12 normal healthy volunteers under fasting conditions. The primary outcome measure was to determine the pharmacokinetics of a buccal dose of TH104, while secondary objectives included establishing the relative bioavailability of TH104 and evaluating its’ tolerability for potential value in clinical efficacy studies.

In this study, the pharmacokinetic evaluation of TH104 transmucosal film compared to an oral tablet marketed in Europe but not the United States, given as an equal-labelled dose in normal healthy volunteers under fasting conditions, was consistent and similar in comparison with results from the literature. The Cmax and AUC0-∞ of TH104 was observed to be higher than the tablet product because of a possible reduced presystemic metabolism in the lower GI and liver, which is potentially advantageous for patients with an impaired liver. The half-life and Tmax was observed to be similar for both products. There were no deaths, other serious adverse events, or other significant adverse events reported during the entire study with events consistent with the safety profile of the marketed tablet in the literature including mild headache and somnolence.
The second phase 1 study was a single-dose, single-center, open-label, parallel group design with 2 treatment groups of 2 different formulations of TH104 with variable pH in sixteen normal healthy volunteers under fasting conditions. The primary outcome measure was to determine the effect of pH in the transmucosal buccal film on drug absorption profile evaluated from serial blood sample collections.
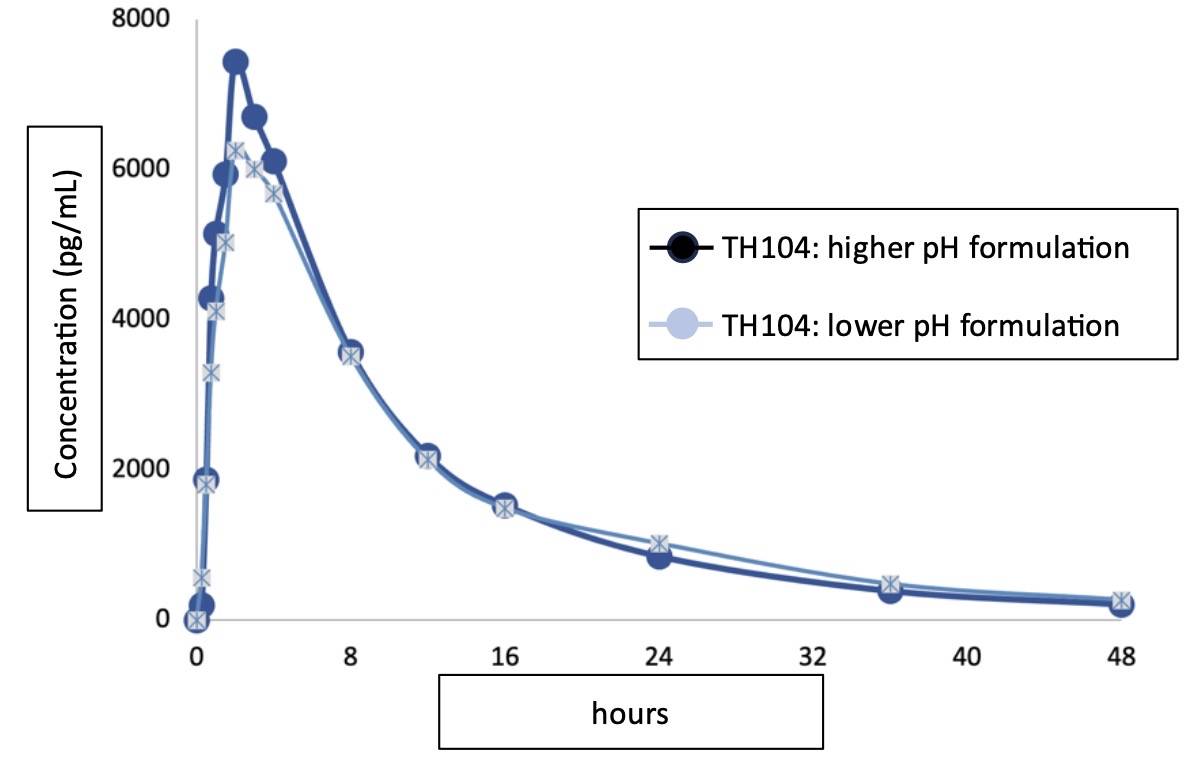
In this study, the evaluation of TH104 transmucosal film given as an equal-labelled dose in 2 different pH formulations delivered buccally to normal healthy volunteers under fasting conditions, had an insignificant effect on the performance of the film as measured by drug concentrations in human plasma. The low impact on pH shows that variations of oral pH due to inter-patient variability in future studies would have a low impact on predictable drug absorption with regards to speed of delivery and drug action once absorbed into the systemic circulation. There were no new adverse events during the entire study with events correlated with the previous study and a safety profile consistent with literature.
The Company intends to provide topline data in 2024 for a phase 1 pharmacokinetic bridging study in the United States designed as a single-dose, single-center, open-label, randomized 2-way crossover study of TH104 transmucosal buccal film and an intravenous dose of drug administered under fasting conditions, with a 7-day washout period between doses. Sixteen normal healthy volunteers will participate in the study. The primary objective is to evaluate the absolute bioavailability of TH104 as well as assess safety and tolerability of the formulation.
Tharimmune believes the completed phase 1 data coupled with the phase 1 pharmacokinetic bridging study in the United States may complement a phase 2a efficacy study to be launched next year in chronic pruritis in PBC as we begin engaging regulatory authorities in both the US and the EU. The phase 2a trial is planned as a multiple ascending dose trial to assess the safety and tolerability of TH104 and will also evaluate the change from baseline in the validated endpoint for pruritis studies, the Worst Itch-Numerical Rating Scale (WI-NRS).
“We are pleased with the Phase 1 data with TH104 which shows reliable and predictable delivery of the active ingredient using our proprietary microparticle embedded transmucosal delivery system which can easily be applied to the inside of the cheek in seconds,” said Randy Milby, CEO of Tharimmune. “We are now poised to continue planning efficacy studies in liver disease patients suffering from debilitating and unrelenting pruritis which many people with PBC deal with on a daily basis with inadequate treatment.”
According to the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK), part of the National Institutes of Health1, PBC, is a chronic disease in where the bile ducts in the liver eventually become dysfunctional and cause the buildup of bile which causes liver damage. The disease, believed to be an autoimmune condition, affects an estimated 58 out of every 100,000 U.S. women and about 15 out of every 100,000 U.S. men. Pruritus is one of the most common conditions associated with PBC affecting up to 75% of individuals at some point during their disease course. It has a negative impact on health-related quality of life with limited treatment options.2 In an on-line survey focusing on certain features of patients’ itch respondents described their itch as “bugs crawling” as well as more than 65% of participants reporting that the itch was worse at night, known as nocturnal pruritis.3
Tharimmune believes TH104 has expansion opportunities in treating chronic pruritogenic conditions associated with cholestatic liver disease as well as other liver-related and non-liver related conditions including fatty and alcoholic hepatic diseases, non-alcoholic liver diseases and certain types of hepatitis. Chronic pruritis is significant in liver diseases as well as chronic kidney diseases, hemodialysis as well as atopic dermatitis.
About TH104
TH104 is embedded with the active pharmaceutical ingredient onto a proprietary transdermal buccal film which easily adheres to the inside of the mouth. This endows TH104 with key features making it an ideal product candidate for multiple liver-related and other pruritogenic inflammatory conditions. The active molecule has a dual mechanism of action affecting both the µ-opioid receptor and the kappa opioid receptor as well as inhibiting IL-17 inflammatory cytokine expression. These opioid receptors when stimulated and/or inhibited by the body’s natural ligands have been known to be involved in the body’s itch circuitry.
About Tharimmune, Inc.
Tharimmune, Inc. is a clinical-stage biotechnology company developing a portfolio of therapeutic candidates for rare, inflammatory, and oncologic diseases. The Company has acquired an exclusive worldwide license for a clinical-stage asset, known to suppress chronic, debilitating pruritis or “uncontrollable itching” in PBC, a rare and orphan liver disease with no known cure. The Company’s early-stage immuno-oncology pipeline includes novel multi-specific antibodies targeting unique epitopes with novel mechanisms of action against well-known, validated targets in multiple solid tumors. Tharimmune has a license agreement with OmniAb, Inc. to access the company’s antibody discovery technology platform against specified targets. For more information please visit: www.tharimmune.com.
Forward Looking Statements
Certain statements in this press release are forward-looking within the meaning of the Private Securities Litigation Reform Act of 1995. All statements, other than statements of historical facts, contained in this press release, including statements regarding Tharimmune’s strategy, future operations, future financial position, projected costs, prospects, plans and objectives of management, are forward-looking statements. The words “anticipate,” “believe,” “continue,” “could,” “depends,” “estimate,” “expect,” “intend,” “may,” “ongoing,” “plan,” “potential,” “predict,” “project,” “target,” “should,” “will,” “would,” and similar expressions are intended to identify forward-looking statements, although not all forward-looking statements contain these identifying words. The Company may not actually achieve the plans, intentions, or expectations disclosed in these forward-looking statements, and you should not place undue reliance on these forward-looking statements. Actual results or events could differ materially from the plans, intentions and expectations disclosed in these forward-looking statements. Factors that may cause such differences, include, but are not limited to, those discussed under Risk Factors set forth in our Annual Report on Form 10-K/A for the year ended December 31, 2022 and other periodic reports filed by the Company from time to time with the Securities and Exchange Commission. In addition, the forward-looking statements included in this press release represent the Company’s views as of the date of this release. Subsequent events and developments may cause the Company’s views to change; however, the Company does not undertake and specifically disclaims any obligation to update or revise any forward-looking statements to reflect new information, future events or circumstances or to reflect the occurrences of unanticipated events, except as may be required by applicable law. These forward-looking statements should not be relied upon as representing the Company’s views as of any date subsequent to the date of this release.
Investor Relations Contact
ir@tharimmune.com
www.tharimmune.com
References
- National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) accessed on November 26, 2023
- Gungabissoon U, Gibbons DC, Requena G, Ribeiro de Souza A, Smith H. Disease burden of primary biliary cholangitis and associated pruritus based on a cross-sectional US claims analysis. BMJ Open Gastroenterol. 2022 Aug;9(1):e000857. doi: 10.1136/bmjgast-2021-000857
- Rishe, E., Azarm, A., & Bergasa, N. V. (2007). Itch in Primary Biliary Cirrhosis: A Patients’ Perspective. Acta Dermato-Venereologica, 88(1), 34-37. doi: 10.2340/00015555-0350
SOURCE: Tharimmune, Inc.
View source version on accesswire.com:
https://www.accesswire.com/810126/tharimmune-reports-positive-phase-1-data-on-th104-its-lead-therapeutic-candidate-for-chronic-pruritis-in-primary-biliary-cholangitis-pbc
