Matinas BioPharma Announces Positive in vivo Safety Data with its Oral LNC-Docetaxel Formulation

Matinas’ Positivein vivo Safety Data with its Oral LNC-Docetaxel Formulation
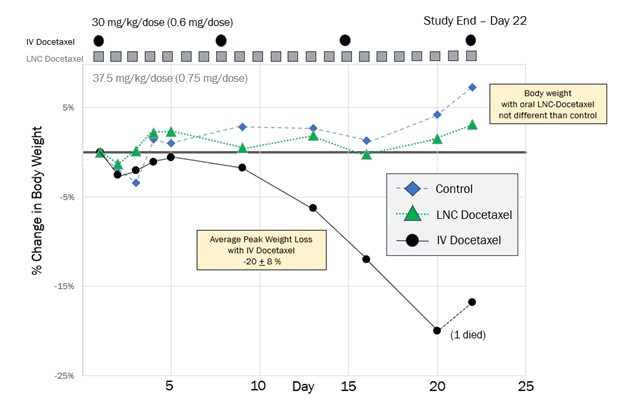
Daily oral LNC-docetaxel, at a total administered dose of more than 8x greater than IV-docetaxel, showed no evidence of weight loss, compared to an average peak weight loss of 20% with IV-docetaxel
Results build on data from prior in vivo study of oral LNC-docetaxel showing reductions in tumor size comparable to IV-docetaxel
Management to host to 2023 Financial Results and Business Update Conference Call on March 27, 2024
BEDMINSTER, N.J., March 25, 2024 (GLOBE NEWSWIRE) — Matinas BioPharma Holdings, Inc. (NYSE American: MTNB), a clinical-stage biopharmaceutical company focused on delivering groundbreaking therapies using its lipid nanocrystal (LNC) platform technology, announces positive results from an additional in vivo study in healthy mice with an oral LNC formulation of docetaxel, a well-established chemotherapeutic agent used in the management of multiple metastatic and unresectable tumors. Docetaxel is currently only administered intravenously and is frequently associated with significant side effects and treatment-limiting toxicities.
“These recent data show how our oral LNC-docetaxel formulation can dramatically reduce the well-recognized toxicity of IV-docetaxel, as primarily manifested by weight loss in this model,” said James J. Ferguson, M.D., Chief Medical Officer of Matinas. “Our previous in vivo study demonstrated that oral LNC-docetaxel effectively targeted melanoma tumors and was able to reduce tumor sizes to a degree comparable to that of IV-docetaxel, with no evident toxicity. We have now corroborated this lack of toxicity in a more comprehensive safety study with a longer treatment duration and even higher doses of oral LNC-docetaxel.
“This illustrates how the crystalline structure of our LNCs encapsulates and protects the body from the docetaxel cargo prior to selectively delivering it directly to tumor cells, markedly reducing the amount of circulating free drug, and thereby avoiding one of the primary drivers of chemotherapy-associated toxicity. Importantly, the unique phosphatidylserine composition of LNCs allows for selective tumor targeting and delivery, making LNC-docetaxel both an effective targeting vehicle and an efficient oral delivery platform for oncology applications,” he added. “With data from our in vivo studies that suggests a broader therapeutic index for oral LNC-docetaxel, we plan to evaluate higher doses in additional tumors – including those that have not historically responded well to docetaxel therapy – as well as potential improvements in the therapeutic indices of other toxic chemotherapies.”
Study Design
The goal of this study was to determine whether an oral LNC formulation of docetaxel could improve the overall safety profile of conventional IV-administered docetaxel. The study included healthy BALB/c mice (n=24) divided into three treatment groups:
- Control animals treated with oral saline.
- IV-docetaxel (30 mg/kg, or 0.6 mg/dose) administered once a week for three weeks.
- Oral LNC-docetaxel (37.5 mg/kg, or 0.75 mg/dose) administered once daily over three weeks.
The primary endpoint was change in body weight over the treatment period.
Findings
Key Takeaways
- Through Day 22, the total amount of docetaxel administered with the oral LNC-docetaxel formulation was more than 8x greater than with IV-docetaxel (the final IV dose was used to measure pharmacokinetics).
- All mice treated with IV-docetaxel lost a significant amount of weight (toxicity), with an average peak loss of 20% of their original body weight.
- Mice treated with oral LNC-docetaxel maintained their body weight, which was statistically no different than the weight of control mice treated with oral saline.
- One mouse in the IV-docetaxel group died prior to the conclusion of the study and was censored from the analysis (last measurement was -32% body weight loss); the curve of the IV-docetaxel group also reflects the expected 7-day recovery following an IV dose of docetaxel.
- The daily administered oral LNC-docetaxel dose was 50% higher, and the total amount of drug administered was 3.5x greater, than the LNC-docetaxel dose administered in a previous study that demonstrated anti-tumor activity comparable to IV-docetaxel in a syngeneic mouse melanoma model.
Conference Call
Matinas BioPharma will report 2023 financial results after market close on Wednesday, March 27, 2024. Matinas management will host an investment community conference call and webcast to discuss financial results and provide a business update that day at 4:30 p.m. Eastern time (1:30 p.m. Pacific time). To participate in the call, please dial 877-484-6065 (Toll-Free) or 201-689-8846 (Toll). The webcast will be available on the IR Calendar page of the Matinas website and will be archived for six months.
About Matinas BioPharma
Matinas BioPharma is a biopharmaceutical company focused on delivering groundbreaking therapies using its lipid nanocrystal (LNC) platform delivery technology.
Matinas’ lead LNC-based therapy is MAT2203, an oral formulation of the broad-spectrum antifungal drug amphotericin B, which although highly potent, can be associated with significant toxicity. Matinas’ LNC platform provides oral delivery of amphotericin B without the significant nephrotoxicity otherwise associated with IV-delivered formulations. Combining comparable fungicidal activity with targeted delivery results in a lower risk of toxicity and potentially creates the ideal antifungal agent for the treatment of invasive fungal infections. MAT2203 was successfully evaluated in the completed Phase 2 EnACT study in HIV patients suffering from cryptococcal meningitis, meeting its primary endpoint and achieving robust survival. MAT2203 will be further evaluated in a single Phase 3 registration trial (the “ORALTO” trial) as an oral step-down monotherapy following treatment with AmBisome® (liposomal amphotericin B) compared with the standard of care in patients with invasive aspergillosis who have limited treatment options.
In addition to MAT2203, preclinical and clinical data have demonstrated that this novel technology can potentially provide solutions to many challenges of achieving safe and effective intracellular delivery of both small molecules and larger, more complex molecular cargos including small oligonucleotides such as ASOs and siRNA. The combination of its unique mechanism of action and flexibility with routes of administration (including oral) positions Matinas’ LNC technology to potentially become a preferred next-generation orally available intracellular drug delivery platform for infection, inflammation and oncology. For more information, please visit www.matinasbiopharma.com.
Forward-looking Statements
This release contains “forward-looking statements” within the meaning of the Private Securities Litigation Reform Act of 1995, including those relating to our business activities, our strategy and plans, the future development of its product candidates, including MAT2203, the Company’s ability to identify and pursue development, licensing and partnership opportunities for its products, including MAT2203, or platform delivery technology on favorable terms, if at all, and the ability to obtain required regulatory approval and other statements that are predictive in nature, that depend upon or refer to future events or conditions. All statements other than statements of historical fact are statements that could be forward-looking statements. Forward-looking statements include words such as “expects,” “anticipates,” “intends,” “plans,” “could,” “believes,” “estimates” and similar expressions. These statements involve known and unknown risks, uncertainties and other factors which may cause actual results to be materially different from any future results expressed or implied by the forward-looking statements. Forward-looking statements are subject to a number of risks and uncertainties, including, but not limited to, our ability to continue as a going concern, our ability to obtain additional capital to meet our liquidity needs on acceptable terms, or at all, including the additional capital which will be necessary to complete the clinical trials of our product candidates; our ability to successfully complete research and further development and commercialization of our product candidates; the uncertainties inherent in clinical testing; the timing, cost and uncertainty of obtaining regulatory approvals; our ability to protect the Company’s intellectual property; the loss of any executive officers or key personnel or consultants; competition; changes in the regulatory landscape or the imposition of regulations that affect the Company’s products; and the other factors listed under “Risk Factors” in our filings with the SEC, including Forms 10-K, 10-Q and 8-K. Investors are cautioned not to place undue reliance on such forward-looking statements, which speak only as of the date of this release. Except as may be required by law, the Company does not undertake any obligation to release publicly any revisions to such forward-looking statements to reflect events or circumstances after the date hereof or to reflect the occurrence of unanticipated events. Matinas BioPharma’s product candidates are all in a development stage and are not available for sale or use.
Investor Contact
LHA Investor Relations
Jody Cain
Jcain@lhai.com
310-691-7100
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/4255be86-7f03-4c05-b464-daf2835468b6

