Positrigo Receives FDA Clearance for Breakthrough Brain PET System

After several years of development, the company’s first dedicated brain PET system comes to market in the US.
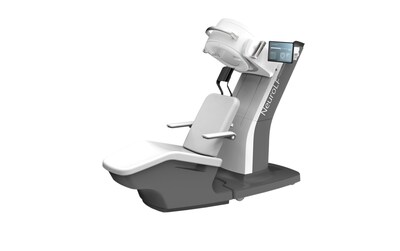
ZURICH, July 16, 2024 /PRNewswire/ — Positrigo, a Swiss based company developing nuclear medical imaging devices to advance functional brain imaging, has achieved a significant milestone with the U.S. Food and Drug Administration (FDA) with the clearance of its dedicated brain PET system NeuroLF®. This innovative imaging device is ultra-compact and assists in diagnosing and monitoring of brain related disorders like Alzheimer’s disease, Brain Tumors, Epilepsy, Parkinson’s disease and others.
“It is not the first device of its kind which receives market clearance in the US but we believe that our patient-centric and customer-driven design and development efforts over the last couple of years, brought us into the pole position to offer the best imaging solution to address the increased demand of brain PET scans,” says Dr. Jannis Fischer, co-founder and CEO. “We are excited to fulfill numerous pre orders in the US and to have first customers benefiting from our technology very soon. It is incredibly rewarding and I couldn’t be prouder of our team which worked tirelessly to achieve this important milestone.”
The NeuroLF system supports a paradigm shift in medical imaging where a dedicated device allows for imaging of a particular body part or organ. What is well established in cardiac (heart) imaging is just starting now for the brain also. By harnessing the advancement in mechanical, electronic and software developments, this system requires only minimal space and no special room modifications, allows patients to receive a brain scan in a seated position and enables functional imaging at the point-of-care.
Increased Demand for Brain PET in the US
The development of a dedicated brain PET device is very timely as just earlier this month another disease-modifying therapy for Alzheimer’s disease received FDA approval[1]. This is already the third medication after two other therapies came to market last year in the US[2]. AD is the most common form of dementia, accounting for ~70% of all dementias in those over 60. In 2020 there were over 55 million people worldwide living with dementia and it is predicted that individuals affected by AD will grow to 139 million in 2050[3]. The early diagnosis is of utmost importance and PET imaging of the brain is essential for the accurate diagnosis of AD patients. However, the currently available PET scanners are large and always combined with MRI or CT. The purchase and maintenance costs are significant and in addition a lot of space is required which limits the availability of these devices.
Commercial traction beyond the US
The growing demand for brain PET in the US was also apparent during the annual meeting of the Society for Nuclear Medicine and Molecular Imaging (SNMMI) this June in Toronto. Representative for others, Prof. Satoshi Minoshima, MD, PhD from University of Utah presented the increased volume in amyloid PET and the PET scanner backlog as seen over the last couple of years[4].
Despite the company’s first market clearance and the very promising customer interest in the US, Positrigo is addressing a global market with growing demand in Europe and Asia. For the European market the regulatory approval is expected for later this year as the required MDR audit has been passed successfully a few weeks ago.
Production is ready to scale
With the existing pre-orders and the strong demand from customers worldwide, the company is gearing up its production capabilities. Dr. Max Ahnen, co-founder and COO is excited about this next step: “Thanks to our excellent partner network we can rely on well-established and experienced companies which are ready to support us in this critical growth stage. I can’t wait to see the first NeuroLF devices leaving our production floor to allow clinicians to offer high quality patient care.”
With the FDA clearance of its first product, Positrigo achieved a major milestone which not only allows the company to generate revenue but brings it one step closer to fulfill its mission – imaging everyone.
About Positrigo:
Positrigo is a pioneer in nuclear medical imaging technologies. Headquartered in Zurich, Switzerland, the medical device company was founded in 2018 as a spin-off of ETH Zurich. Positrigo’s technology, development, clinical testing and commercialization has been supported by various private investors, the Swiss government and the European Innovation Council. NeuroLF – the company’s first device – is an ultra-compact brain Positron Emission Tomography (PET) scanner which has applications in the assessment of causes of dementias, such as Alzheimer’s disease and other brain related disorders. Learn more at http://www.positrigo.com.
Images:
[1] NeuroLF System
[2] Positrigo Logo
References:
[1] Kisunla (Donanemab) from Eli Lilly receives FDA approval (July 2024
[2] Traditional Approval of Lecanemab (07/2023)
[3] World Alzheimer Report 2022
[4] 2024 SNMMI Annual Meeting Program
Photo – https://healthtechnologynet.com/wp-content/uploads/2024/07/Positrigo_AG.jpg
Logo – https://healthtechnologynet.com/wp-content/uploads/2024/07/Positrigo_AG_Logo.jpg
![]() View original content to download multimedia:https://www.prnewswire.com/news-releases/positrigo-receives-fda-clearance-for-breakthrough-brain-pet-system-302197969.html
View original content to download multimedia:https://www.prnewswire.com/news-releases/positrigo-receives-fda-clearance-for-breakthrough-brain-pet-system-302197969.html
SOURCE Positrigo AG