Hemostemix is Up-Listing in the USA to Generate Liquidity

Calgary, Alberta–(Newsfile Corp. – January 2, 2025) – Hemostemix Inc. (TSXV: HEM) (OTC: HMTXF) (FSE: 2VF0) (“Hemostemix” or the “Company”) is pleased to announce it has applied to up-list its shares on the OTCQB Venture Market. Currently listed on the TSXV Exchange, Frankfurt, and the OTC, Hemostemix aims to increase its liquidity by broadening its reach to all investors who trade in the capital markets of the USA, Europe, and Canada.
The Company’s social media campaign across Instagram, Facebook, LinkedIn, YouTube, and X, describe the patients’ treatment results in their own words. Individual patients describe how the Hemostemix therapy, ACP-01, saved their limb from amputation, regenerated their heart function, successfully treated congestive heart failure, and overcame vascular dementia. These outcomes highlight Hemostemix as an investment opportunity.
“Our testimonials connect ACP-01-treated with the millions of no-option patients who may obtain an ACP treatment under special access program,” stated Thomas Smeenk, CEO. “For example, 236 million suffer from peripheral arterial disease. 23 million degenerate into chronic limb threatening ischema (“CLTI”), and face a 60% mortality rate in five years. Whereas the University of Toronto and University of British Columbia posted their interim results during our Phase II randomized double blind clinical trial that 83% of patients followed for up to 4.5 years experienced healing of ulcers and resolution of ischemic rest pain. Look at the evidence below,” Smeenk said.
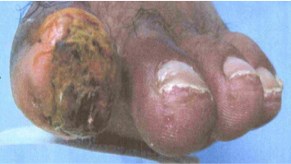
Before Treatment
Cannot view this image? Visit:
https://healthtechnologynet.com/wp-content/uploads/2025/01/235778_6910c01b4421da8b_002full.jpg
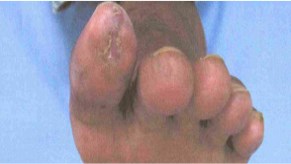
After Treatment with Hemostemix ACP-01
Cannot view this image? Visit:
https://healthtechnologynet.com/wp-content/uploads/2025/01/235778_6910c01b4421da8b_004full.jpg
Precision Healthcare (ACP-01) Restores Quality of Life
ACP-01 is sourced from the patient’s blood and cultured in the patient’s serum. It is demonstrated to be safe and statistically effective. For example:
-
In dilated cardiomyopathy patients, cardiac function as measured by left ventricle ejection fraction percent (LVEF%) increased by up to 47.1% following one treatment (Stem Cell Research & Therapy, November 2023), with the most marked improvements observed in patients with severe dilated cardiomyopathy (LVEF% < 20%).
-
The Company’s published phase II results detailed that ulcer size decreased significantly in the treated group from a mean of 1.46 cm² to 0.48 mm², p = 0.01 within three months (Journal of Biomedical Research & Environmental Science, February 2024).
Follow Hemostemix:
- Instagram: https://www.instagram.com/hemostemix.inc?utm_source=ig_web_button_share_sheet&igsh=ZDNlZDc0MzIxNw==
- Facebook: https://www.facebook.com/profile.php?id=61571076998595
- LinkedIn: https://www.linkedin.com/in/thomas-smeenk-03b9213/
- YouTube: https://www.youtube.com/@hemostemix3830
- X: @heartrepairguy
About Hemostemix
Founded in 2003, Hemostemix is a clinical-stage biotechnology company and winner of the World Economic Forum Technology Pioneer Award. The Company has developed and patented a blood-based stem cell therapeutics platform that includes angiogenic cell precursors (ACP), neuronal cell precursors (NCP), and cardiomyocyte cell precursors (CCP)
For more information, visit www.hemostemix.com.
For further information, please contact:
Thomas Smeenk, President & CEO
Email: tsmeenk@hemostemix.com
Phone: 905-580-4170
Neither the TSX Venture Exchange nor its Regulation Service Provider (as that term is defined under the policies of the TSX Venture Exchange) accepts responsibility for the adequacy or accuracy of this release.
Forward-Looking Information: This news release contains “forward-looking information” within the meaning of applicable Canadian securities legislation. All statements, other than statements of historical fact, included herein are forward-looking information. In particular, this news release contains forward-looking information in relation to sales of its lead product ACP-01, the commercialization of ACP-01 via the sale of compassionate treatments under Special Access Program, and social media programs targeted to generate sales. There can be no assurance that such forward-looking information will prove to be accurate. Actual results and future events could differ materially from those anticipated in such forward-looking information. This forward-looking information reflects Hemostemix’s current beliefs and is based on information currently available to Hemostemix and on assumptions Hemostemix believes are reasonable. These assumptions include, but are not limited to: the underlying value of Hemostemix and its Common Shares; the successful resolution of the litigation that Hemostemix is pursuing or defending (the “Litigation“); the results of ACP-01 research, trials, studies and analyses, including the analysis being equivalent to or better than previous research, trials or studies; the receipt of all required regulatory approvals for research, trials or studies; the level of activity, market acceptance and market trends in the healthcare sector; the economy generally; consumer interest in Hemostemix’s services and products; competition and Hemostemix’s competitive advantages; and Hemostemix obtaining satisfactory financing to fund Hemostemix’s operations including any research, trials or studies, and any Litigation. Forward-looking information is Subject to known and unknown risks, uncertainties and other factors that may cause the actual results, level of activity, performance or achievements of Hemostemix to be materially different from those expressed or implied by such forward-looking information. Such risks and other factors may include, but are not limited to: the ability of Hemostemix to complete clinical trials, complete a satisfactory analyses and file the results of such analyses to gain regulatory approval of a phase II or phase III clinical trial of ACP-01; potential litigation Hemostemis may face; general business, economic, competitive, political and social uncertainties; general capital market conditions and market prices for securities; delay or failure to receive board or regulatory approvals; the actual results of future operations including the actual results of future research, trials or studies; competition; changes in legislation affecting Hemostemix; the timing and availability of external financing on acceptable terms; long-term capital requirements and future developments in Hemostemix’s markets and the markets in which it expects to compete; lack of qualified, skilled labour or loss of key individuals; and risks related to the COVID-19 pandemic including various recommendations, orders and measures of governmental authorities to try to limit the pandemic, including travel restrictions, border closures, non-essential business closures service disruptions, quarantines, self-isolations, shelters-in-place and social distancing, disruptions to markets, disruptions to economic activity and financings, disruptions to supply chains and sales channels, and a deterioration of general economic conditions including a possible national or global recession or depression; the potential impact that the COVID-19 pandemic may have on Hemostemix which may include a decreased demand for the services that Hemostemix offers; and a deterioration of financial markets that could limit Hemostemix’s ability to obtain external financing. A description of additional risk factors that may cause actual results to differ materially from forward-looking information can be found in Hemostemix’s disclosure documents on the SEDAR website at www.sedarplus.ca. Although Hemostemix has attempted to identify important factors that could cause actual results to differ materially from those contained in forward-looking information, there may be other factors that cause results not to be as anticipated, estimated or intended. Readers are cautioned that the foregoing list of factors is not exhaustive. Readers are further cautioned not to place undue reliance on forward-looking information as there can be no assurance that the plans, intentions or expectations upon which they are placed will occur. Forward-looking information contained in this news release is expressly qualified by this cautionary statement. The forward-looking information contained in this news release represents the expectations of Hemostemix as of the date of this news release and, accordingly, it is Subject to change after such date. However, Hemostemix expressly disclaims any intention or obligation to update or revise any forward-looking information, whether as a result of new information, future events or otherwise, except as expressly required by applicable securities law.
To view the source version of this press release, please visit https://www.newsfilecorp.com/release/235778
