Neurocare Group Receives FDA Clearance for Its Industry-Leading Intermittent Theta Burst Stimulation (iTBS) Protocol for Transcranial Magnetic Stimulation (TMS)

neurocare is pleased to announce that it has received FDA clearance for its industry-leading intermittent Theta Burst Stimulation (iTBS) protocol, available across its Apollo TMS Therapy System and other Transcranial Magnetic Stimulation systems for clinical practice.
ATLANTA, GA / ACCESSWIRE / August 22, 2024 / neurocare group (“neurocare” or the “Company”), a leading innovator in personalized mental health and performance, is pleased to announce that it has received FDA clearance for its industry-leading intermittent Theta Burst Stimulation (iTBS) protocol, available across its Apollo TMS Therapy System and other Transcranial Magnetic Stimulation systems for clinical practice.
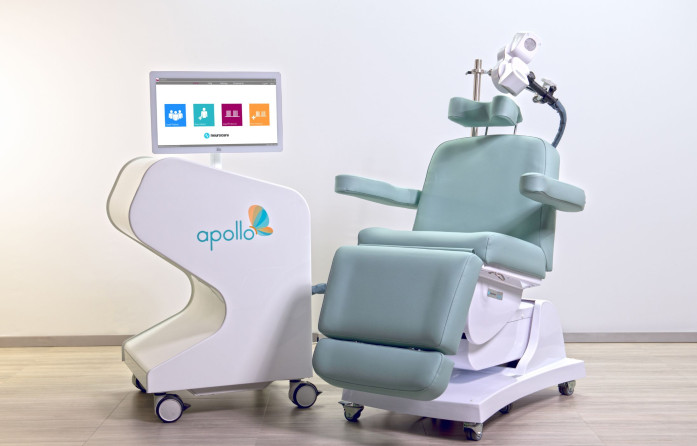
TMS is a non-invasive brain stimulation technique and an evidence-based treatment for depression which can be delivered in ambulant settings as an alternative to medications and with minimal side-effects. During TMS therapy, a magnetic coil on the patient’s head emits a focal stimulation to boost neural pathways in the brain.
iTBS is a more time-efficient treatment protocol with just over three minutes required for an individual treatment session for patients with depression. Additionally, this allows for more sophisticated treatment protocols, giving the provider more flexibility in treatment to enhance clinical care.
In recent years, neurocare and its TMS team in Germany have engineered an advanced technology for delivering iTBS which ensures a consistent and efficient delivery of full stimulation power across the entire burst sequence. This offers a superior solution to other TMS systems on the market, where the pulse strength can decay with subsequent pulses, resulting in less than optimal clinical care.
“We knew it was important to give physicians the confidence in using innovative technologies like TMS, so it was a key focus area for our team to find a solution to the challenge of output power consistency in iTBS stimulation,” said Dr. Kerstin Häringer, Group Head of TMS technology at neurocare group, Munich, Germany. “Thanks to the dedication of our world-class engineering team, we achieved superior power consistency for our TMS systems, thereby making the iTBS protocol more reliable.”
When being treated with an iTBS protocol on the Apollo TMS system, a patient can now receive a reliable stimulation dose that accurately reflects the parameters set by the physician.
“We are excited to be able to offer this new best-in-class technology so our customers can in turn provide the best care to their patients,” said Stanford Miller, Group Executive Vice President, and Managing Director of neurocare group North American operations.
Since 2021, neurocare has built a substantial network of clinic partnerships and private physician users in the field of psychiatry across the USA. More than 175 clinics and institutions have adopted the Apollo TMS Therapy system to help the most difficult-to-treat patients they service. The Apollo is FDA-cleared as a treatment for major depression after a patient has tried and not benefited from oral antidepressant medications. Apollo TMS Therapy is a covered service by most private and public health insurance providers.
Contact Information
Sally Remington
Global Marketing Manager
media@neurocaregroup.com
+49 152 2305 8385
SOURCE: neurocare group
View the original press release on newswire.com.
